<<PREVIOUS -
HOME -
CONTENTS -
NEXT>>
SPECTROSCOPY INDEX -
![]()
<<PREVIOUS -
HOME -
CONTENTS -
NEXT>>
SPECTROSCOPY INDEX -
![]()
|
Screwdriver CD spectroscope is built with a screwdriver that has a round bright shaft and a piece of CD that has its label removed. The CD is mounted in a tube. The screwdriver round bright shaft is used as the slit to reflect a sliver of light towards the CD. The clear CD is used as a transmission diffraction grating to disperse the light into a spectrum. Construction Details The CD label was removed by an air compressor or you can use tape (by niethammerzoltan). To see the spectrum better the CD is mounted in the end of a three inch cardboard tube. To improve the spectrum contrast the tube is made darker on the inside by putting a half inch slot in the other end of the tube. Also, black felt is rolled into a cylinder and put in to the tube to decrease light reflections off the inside of the tube. In addition, three black foam light field stops (diaphragms) about 2 inches in diameter were put in the tube to reduce light reflections off the inside of the tube. The CD is aligned with the half inch slot so that the spectrum is on each side of the wide slot. The tube is mounted on the camera tripod so it is steady and it is confortable to look through. Using the Screwdriver CD Spectroscope The screwdriver is put on the ground or held by a small vice to reflect the Sun’s light into the tube. Black felt is put behind the screwdriver to increase the light contrast. The screwdriver CD spectroscope is easy to use visually. The hydrogen beta, magnesium triple and sodium doublet absorption lines are easily observed.
Taking a photo of the spectrum is challenge. A tripod is used to position the camera over the CD. The camera needs to focus close in to obtain a sharp image of the spectrum. |

|

|

|

|

|
|
Sun Spectrum
Warning The solar spectrum image was taken with the screwdriver CD spectroscope and a Nikon D70 DSLR with a manual Nikkor 55mm macro f/2.8 lens at f/8. The focus was adjusted for the green part of the spectrum to capture the magnesium triplet. The three magnesium triplet absorption lines are seen in the image. The dark blue absorption line is hydrogen beta and the thick double dark line in the yellow-orange is the sodium doublet. The short wavelengths are on the left side of the spectrum which is blue and the long wavelengths are on the right side of the spectrum which is red. The far ends of the spectrum are out of focus.
When taking a photo of the spectrum the challenge is to not over expose parts of the spectrum. |

|
|

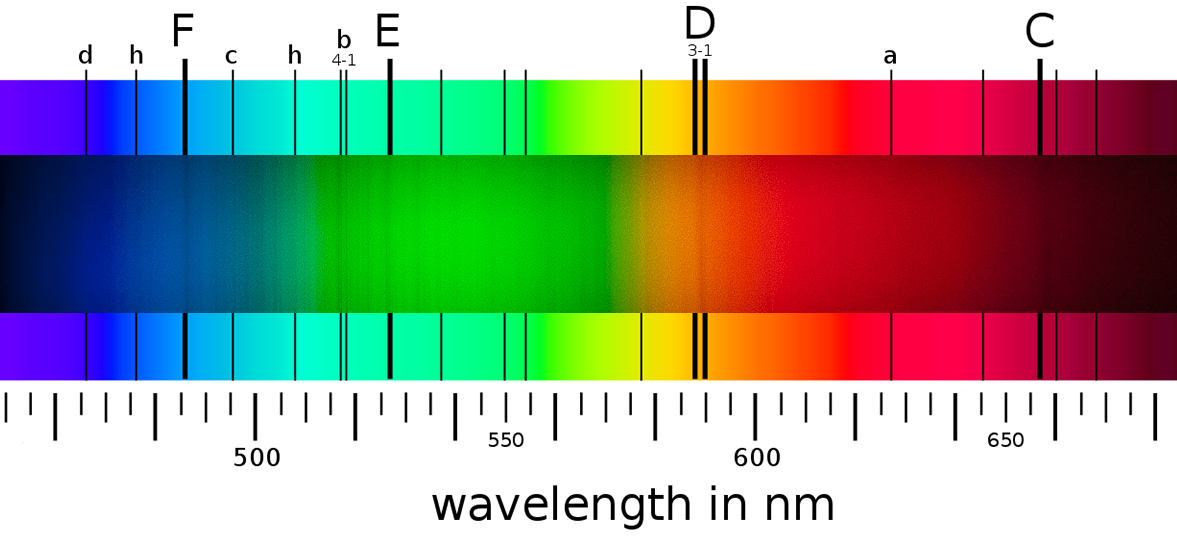
The spectroscope eaisly shows the Fraunhofer absorption lines for F, b4-1, E, D3-1 and C. Fraunhofer absorption lines chart is from Wikipedia with a slice of the solar spectrum image overplayed on it. The spectrum photo is not linear and does not fit exactly on the Fraunhofer absorption lines chart. Dark Fraunhofer absorption lines identified in the spectrum image.
Spectrum Anaylsis from Hydrogen Beta to Sodium Doublet
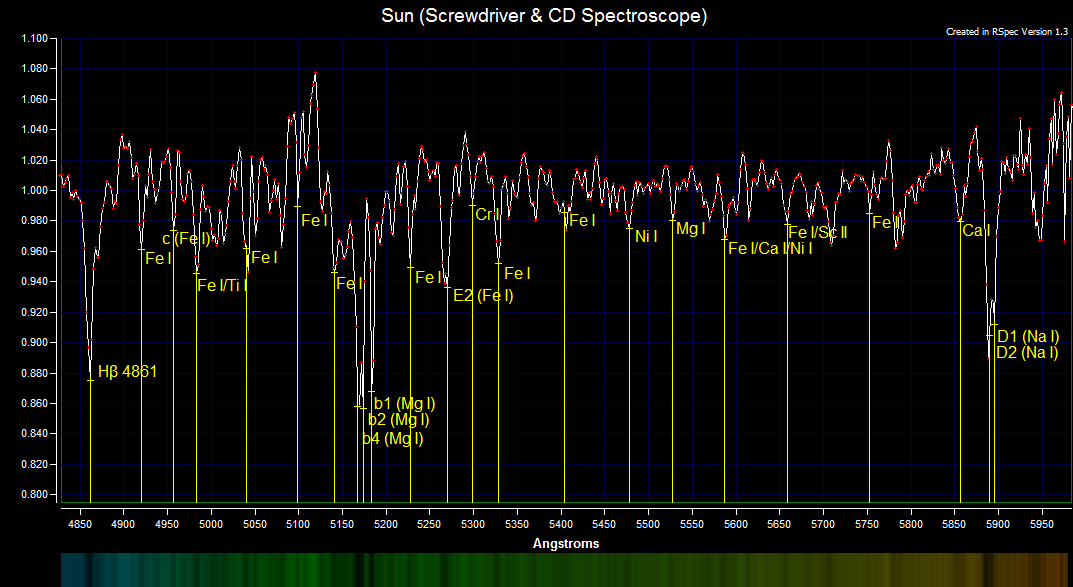 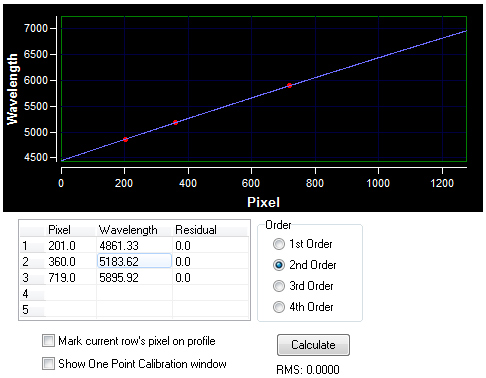
The above spectrum profile is the solar spectrum (uncalibrated intensity data) that has been divided by the shape of its own continuum to remove the continuum. The solar spectrum was wavelength calibrated using RSpec 2nd order on hydrogen beta, the magnesium triplet and the sodium doublet. The difference between the CCD camera pixels at the magnesium triplet wavelength is 2.01 Å. It is impressive that a simple screwdriver and CD shows so many dim absorption lines that match correctly to the elements and resolves the magnesium triplet and the sodium doublet. Solar Spectrum
Solar Hydrogen β Spectrum
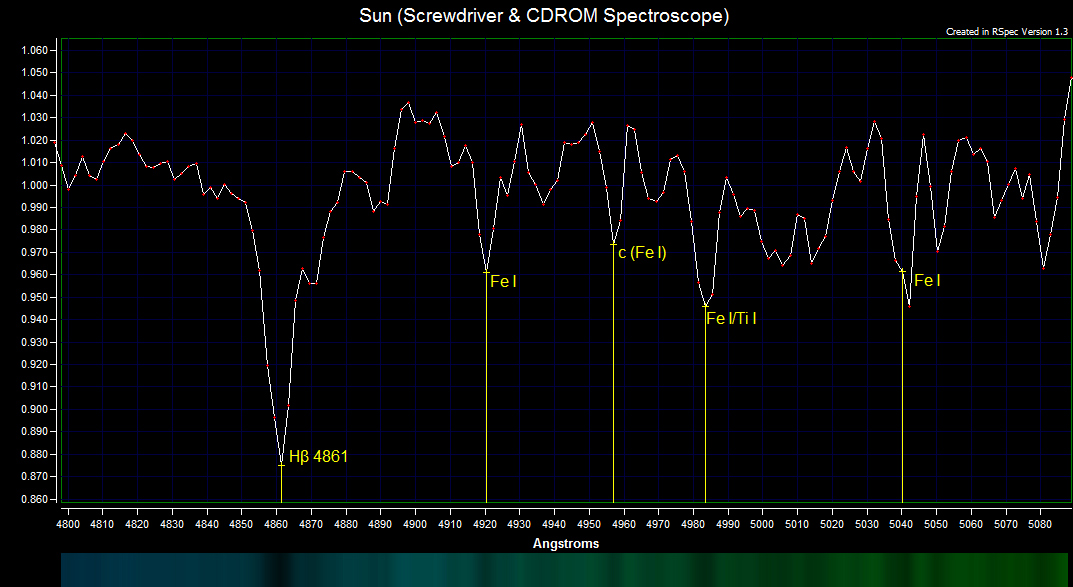 The hydrogen β absorption line contains three strong H I β lines that merge into one line.
Hydrogen Beta Spectrum Energy Levels 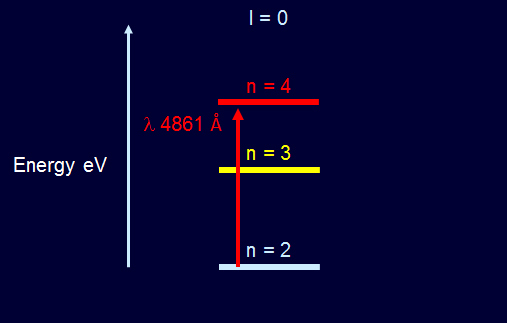 Solar Sodium Doublet Spectrum
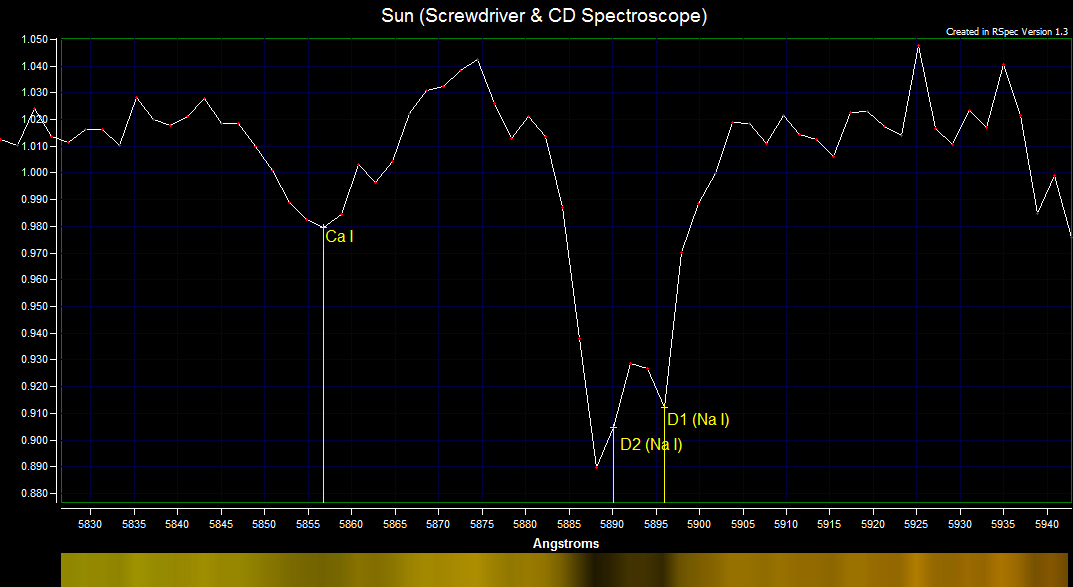 The sodium doublet contains two strong lines of sodium.
The sodium doublet is caused by the absorption of light at λ 5890 Å and λ 5896 Å. The sodium atom absorbs the light and the sodium electron jumps from the n=3, l=0 level to the n=3, l=1 s=+½ level or n=3, l=1 s=-½ level. Sodium Doublet Spectrum Energy Levels 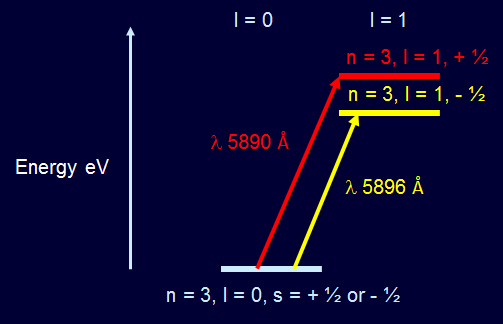 Solar Magnesium Triplet Spectrum
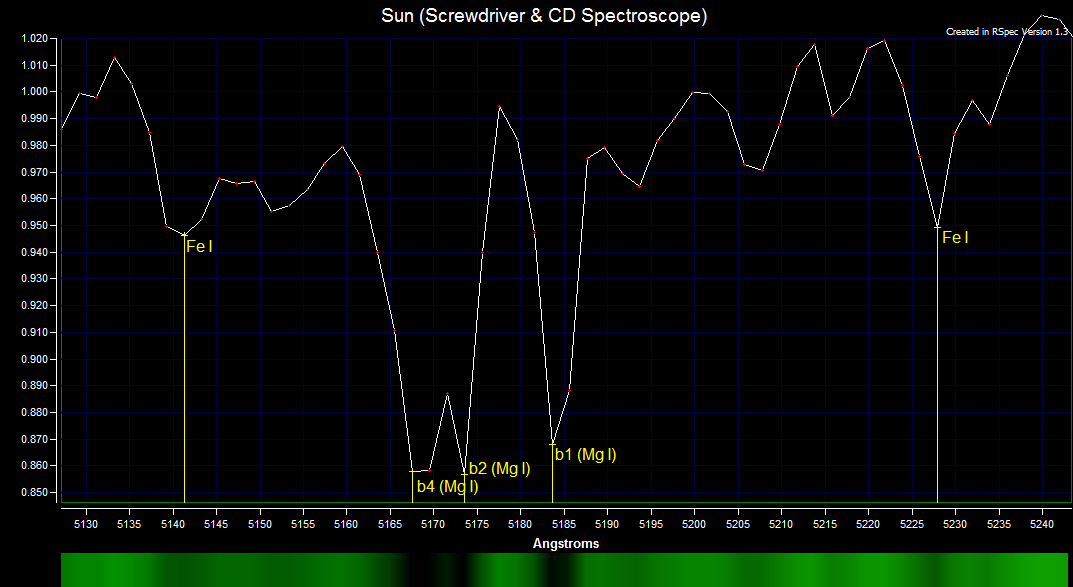 The magnesium triplet contains three strong lines of magnesium and one strong line of iron. There are weaker lines of iron that are not resolved in the magnesium triplet. The b4 absorption line is composed of Mg I 5167.322 Å and Fe I 5167.4883 Å and is wider than the b2 Mg I 5172.684 Å and b1 Mg I 5183.604 Å absorption lines.
Web Pages with Solar Magnesium Triplet and Sodium Doublet Spectrums
Atomic Structure and Spectroscopy
|